Home
Appropriate Patients


76.6%
of patients would prefer to switch to oral
treatment when offered alongside other options2
A forecast of predicted treatment choices among patients with MDS found2:
STUDY DESIGN: A noninterventional, cross-sectional, mixed-methods study of patients with MDS using qualitative and quantitative methods to develop a survey and analyze responses. The objective of the study was to show preferences of patients with MDS in the United States and Canada for hypomethylating agents' benefits, risks, and burden of administration through an online and discrete-choice experiment. Statistical significance was defined with a threshold of 5% of type I error. The survey was completed by 184 of the 275 individuals who initially responded to the invitation.2
Some require travel to and from chemotherapy infusion centers or hospitals for IV infusions or SC injections

Venous access and parenteral administration4-6

Of 664 higher-risk patients, 295 (44.4%) were nonpersistent with HMA treatment (nonpersistence is defined by the investigators as <4 cycles or a gap of ≥90 days between cycles)6

*Surveillance, Epidemiology, and End Results (SEER) Medicare-linked database between 2010 and 2016.


INQOVI tablets are 1 pill taken once daily for 5 days out of a 28-day cycle. See full dosing information here. INQOVI is a fixed-dose combination of decitabine (35 mg) and cedazuridine (100 mg), a cytidine deaminase (CDA) inhibitor that enhances oral bioavailability of decitabine and increases its systemic exposure.
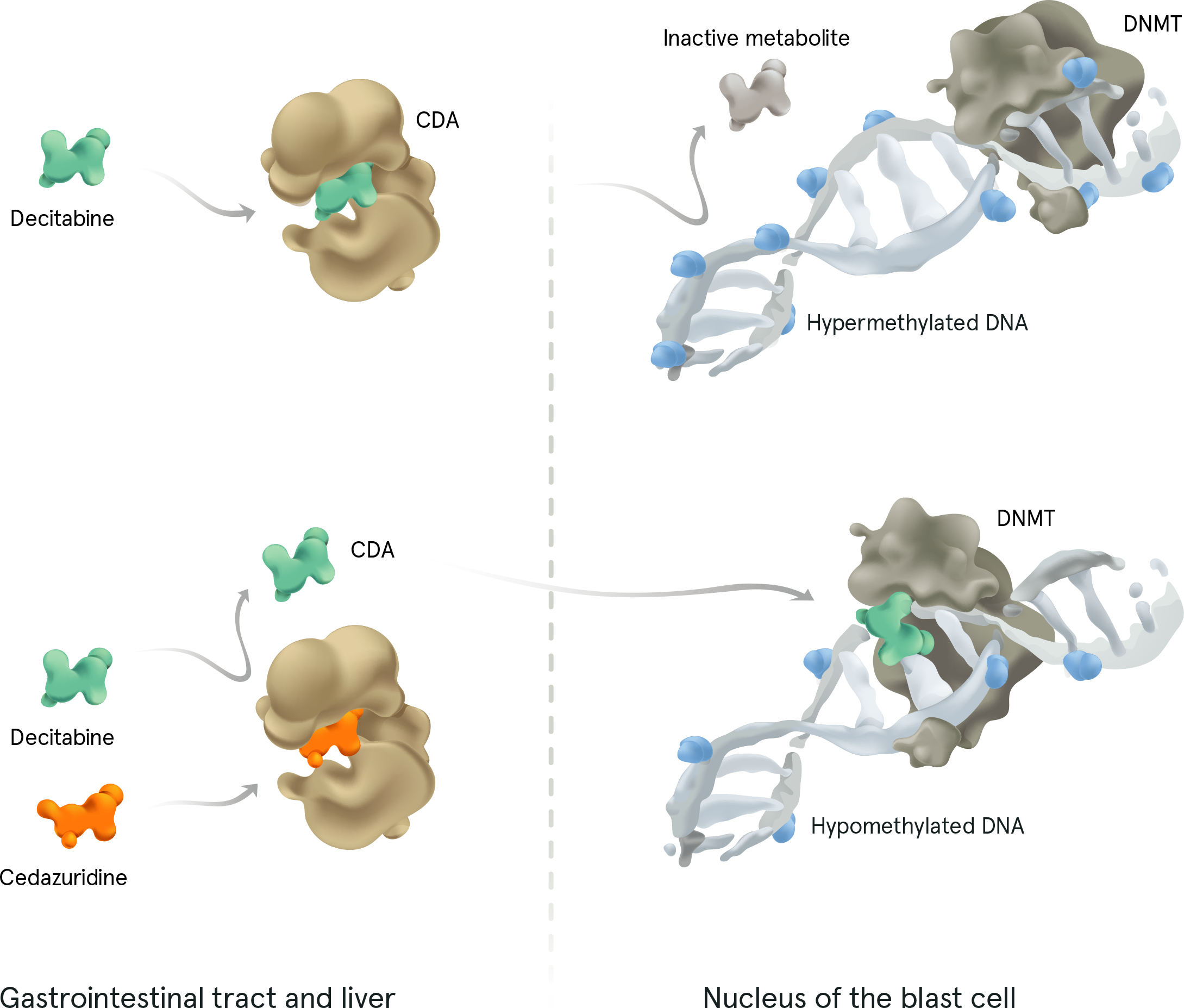
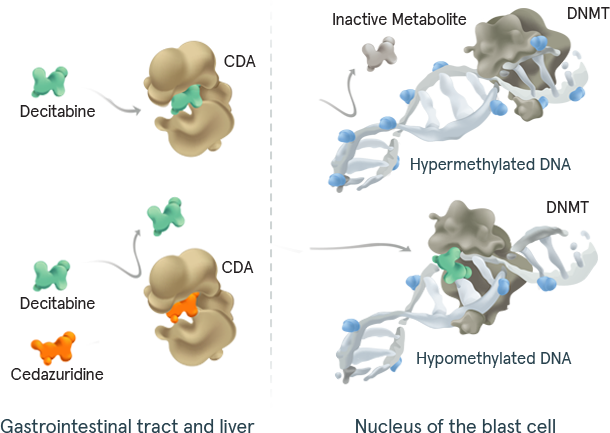
CMML=chronic myelomonocytic leukemia; DNMT=DNA methyltransferase; HMA=hypomethylating agent; IPSS-R=Revised International Prognostic Scoring System; MDS=myelodysplastic syndromes.
References: 1. INQOVI [package insert]. Princeton, NJ: Taiho Oncology, Inc.; 2022. 2. Zeidan AM, Tsai J-H, Karimi M, et al. Patient preferences for benefits, risks, and administration route of hypomethylating agents in myelodysplastic syndromes. Clin Lymphoma Myeloma Leuk. 2022;22(9):e853-e866. 3. Savona MR, Odenike O, Amrein PC, et al. An oral fixed-dose combination of decitabine and cedazuridine in myelodysplastic syndromes: a multicentre, open-label, dose-escalation, phase 1 study. Lancet Haematol. 2019;6(4): e194-e203. 4. Steensma DP, Komrokji RS, Stone RM, et al. Disparity in perceptions of disease characteristics, treatment effectiveness, and factors influencing treatment adherence between physicians and patients with myelodysplastic syndromes. Cancer. 2014;120(11):1670-1676. 5. Leveque D. Subcutaneous administration of anticancer agents. Anticancer Res. 2014;34(4):1579-1586. 6. Joshi N, Kale H, Corman S, et al. Direct medical costs associated with treatment nonpersistence in patients with higher-risk myelodysplastic syndromes receiving hypomethylating agents: a large retrospective cohort analysis. Clin Lymphoma Myeloma Leuk. 2021;21(3):e248-e254. 7. Kini V, Ho PM. Interventions to improve medication adherence: a review. JAMA. 2018;320(23):2461-2473.
Home
Appropriate Patients